Frequently Asked Questions about COVID-19
COVID-19 is a contagious disease caused by the SARS-CoV-2 virus. Most people with COVID-19 have mild symptoms, but some may become severely ill.
Vaccination
Updated and reviewed May 2026
Sources: American Academy of Pediatrics, American College of Obstetricians and Gynecologists, Infectious Diseases Society of America, Centers for Disease Control and Prevention, Food and Drug Administration
Related Resources
2025 National Survey on Respiratory Diseases
The 2025 national survey examines attitudes and behaviors regarding flu, RSV, COVID-19, and pneumococcal disease.
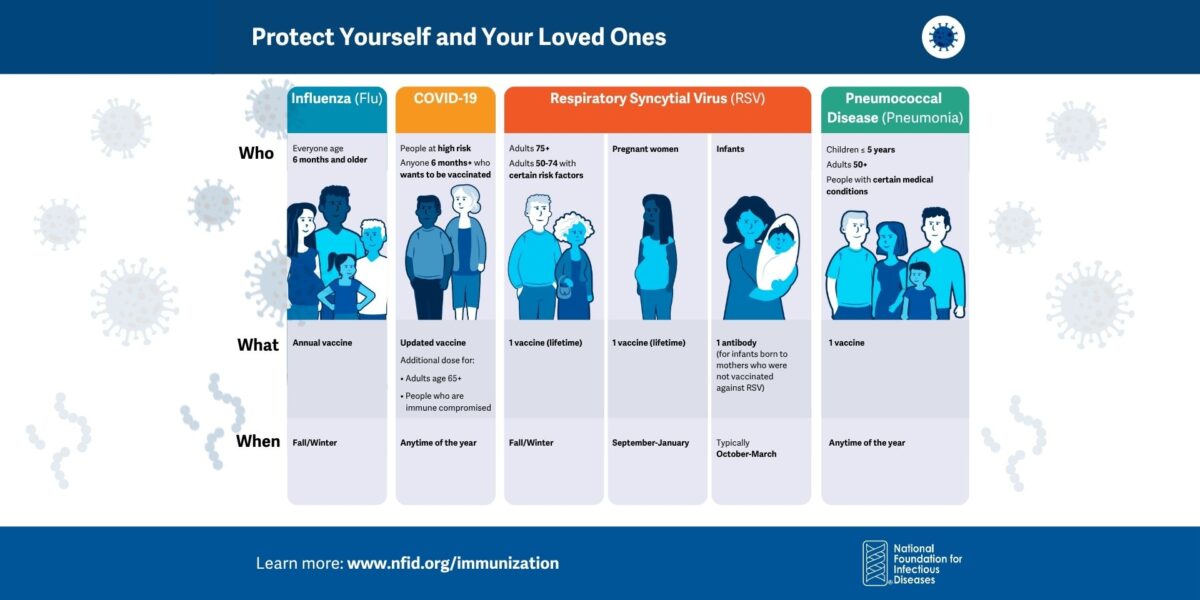
Respiratory Immunization Graphics
Graphics and sample social posts to help raise awareness about preventing COVID-19, flu, RSV, and pneumococcal disease
What Is an Antiviral?
Overview of antivirals—what they are, how they work, and what diseases they help treat